FDA Warns Public Against Fake Version of this Drug Product
The Food and Drug Administration (FDA) has released FDA Advisory No. 0103, series of 2021, reminding the public and healthcare professionals not to purchase and use this verified counterfeit drug product:
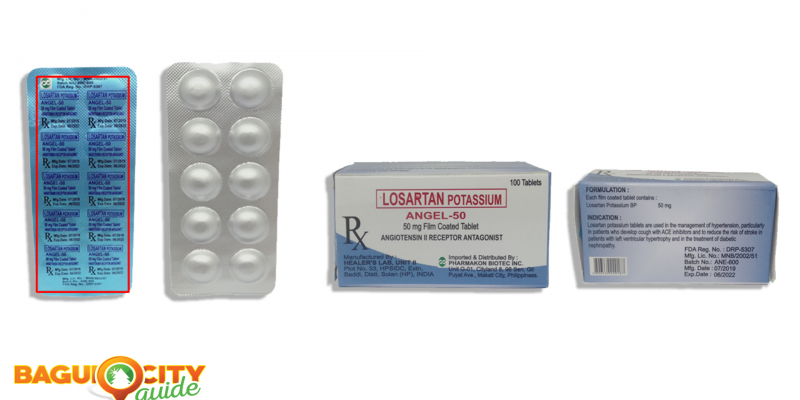
- Losartan Potassium (Angel-50) 50mg Film Coated Tablet
The said counterfeit drug product was verified by FDA and the Marketing Authorization Holder (MAH), Pharmakon Biotec Inc.

Photo Source: FDA
Authentic VS Counterfeit
Take a look at the difference between authentic and verified counterfeit Losartan Potassium (Angel-50) 50mg Film Coated Tablet:
In this first figure, according to FDA, the blister packaging of the counterfeit version has different pantone color and fonts than that of the authentic drug product.

Screenshot from: FDA
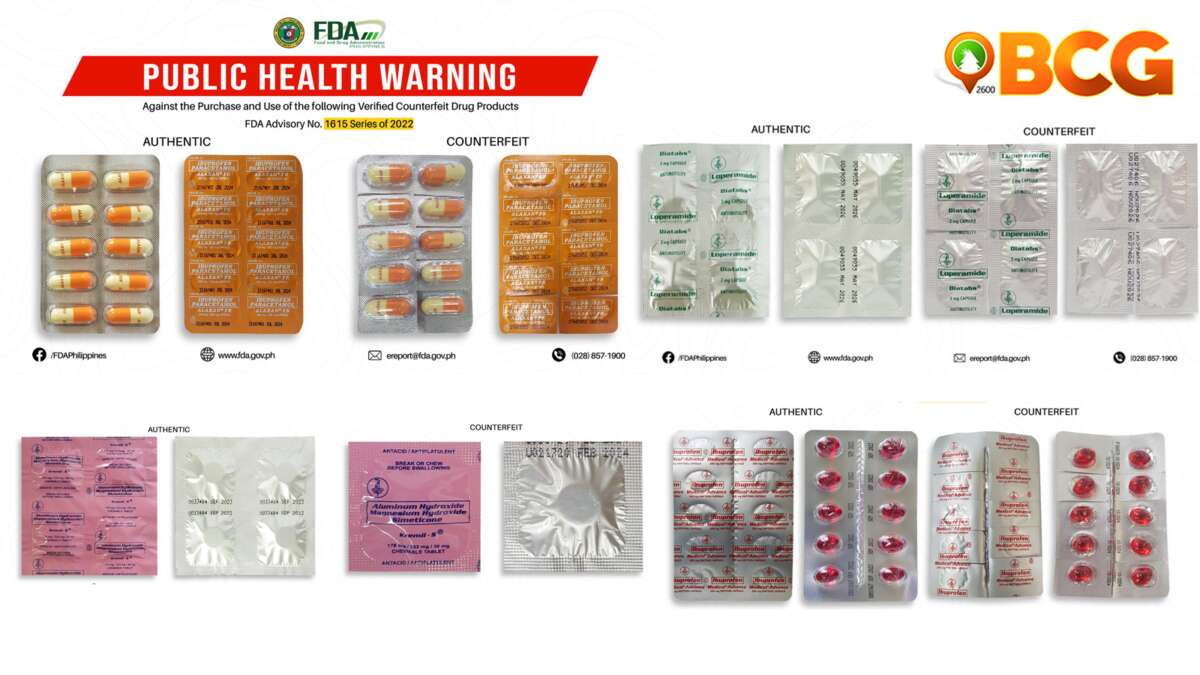
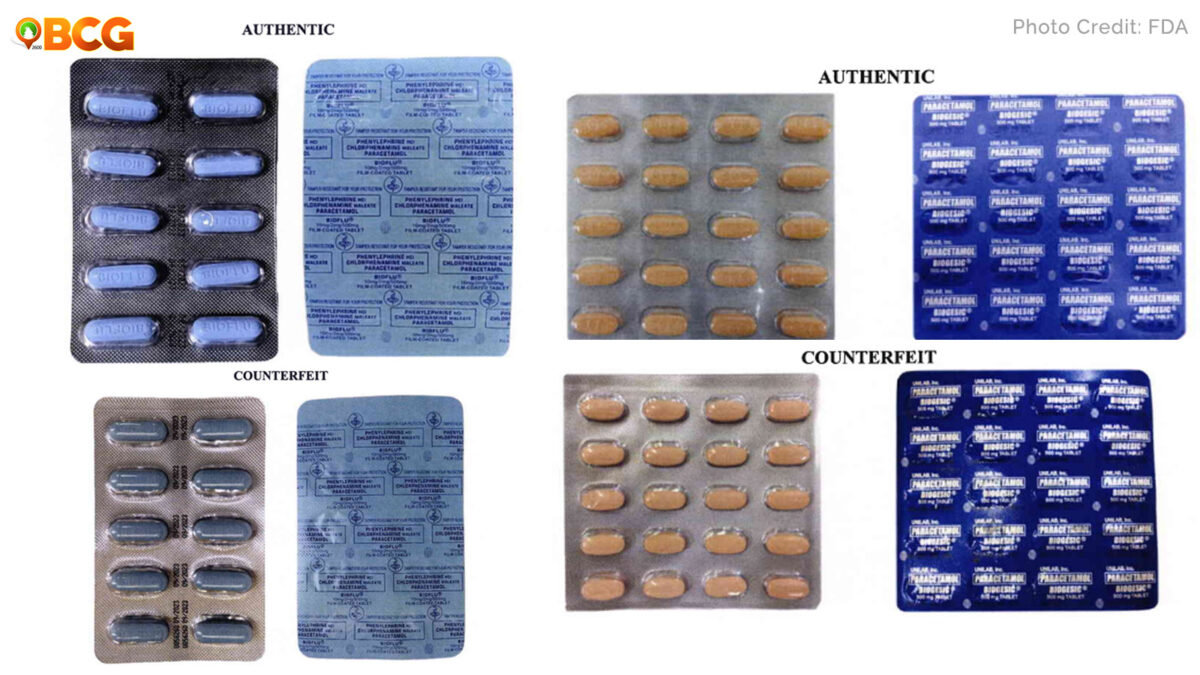
As shown by FDA in this second figure, the Adverse Drug Reaction statement boldness is different.

Screenshot from: FDA
As stated by FDA, everyone is reminded to purchase drug products only from FDA-licensed establishments.
In addition, the importation, selling, or offering for sale of a verified counterfeit drug product is in direct violation of:
- Republic Act No. 9711 or the Food and Drug Administration Act of 2009
- Republic Act No. 8203 or the Special Law on Counterfeit Drugs
Furthermore, anyone found selling the said counterfeit drug product will be penalized according to FDA.
Report Sale/Distribution
To report any sale or distribution of verified counterfeit drug products, report through the online reporting facility eReport.
Related Article:
FDA Warns Public Against Purchase and Use of the Following Unregistered Drug Products
For More News and Updates
Looking for more news and updates? Feel free to explore our BCG website and our official Facebook page. You may also check out our official BCG YouTube channel to catch a variety of video content.
Source: FDA